The presence of viable bacteria in the blood, i.e., bacteremia, when not controlled properly can lead to bloodstream infection (BSI) and sepsis, a syndromic inflammatory response.
Rapid and accurate antimicrobial prescriptions are critical to decrease mortality significantly for BSI patients. However,the traditional antimicrobial susceptibility testing (AST) for BSI is time-consuming and tedious, leading clinicians to rely primarily on experience for treatment.
Researchers from Shandong University, Qingdao Institute of Bioenergy and Bioprocess Technology (QIBEBT) of the Chinese Academy of Sciences (CAS) and the Affiliated Hospital of Qingdao University have developed an integrated microfluidic chip (BSI-AST chip) for rapid AST from positive blood cultures (PBCs) and the whole process from bacteria extraction to AST result output was less than 3.5 hours.It promises to be a powerful new tool for accelerating AST for bloodstream infections.
The study was published in Analytical Chemistry on Sept. 14.
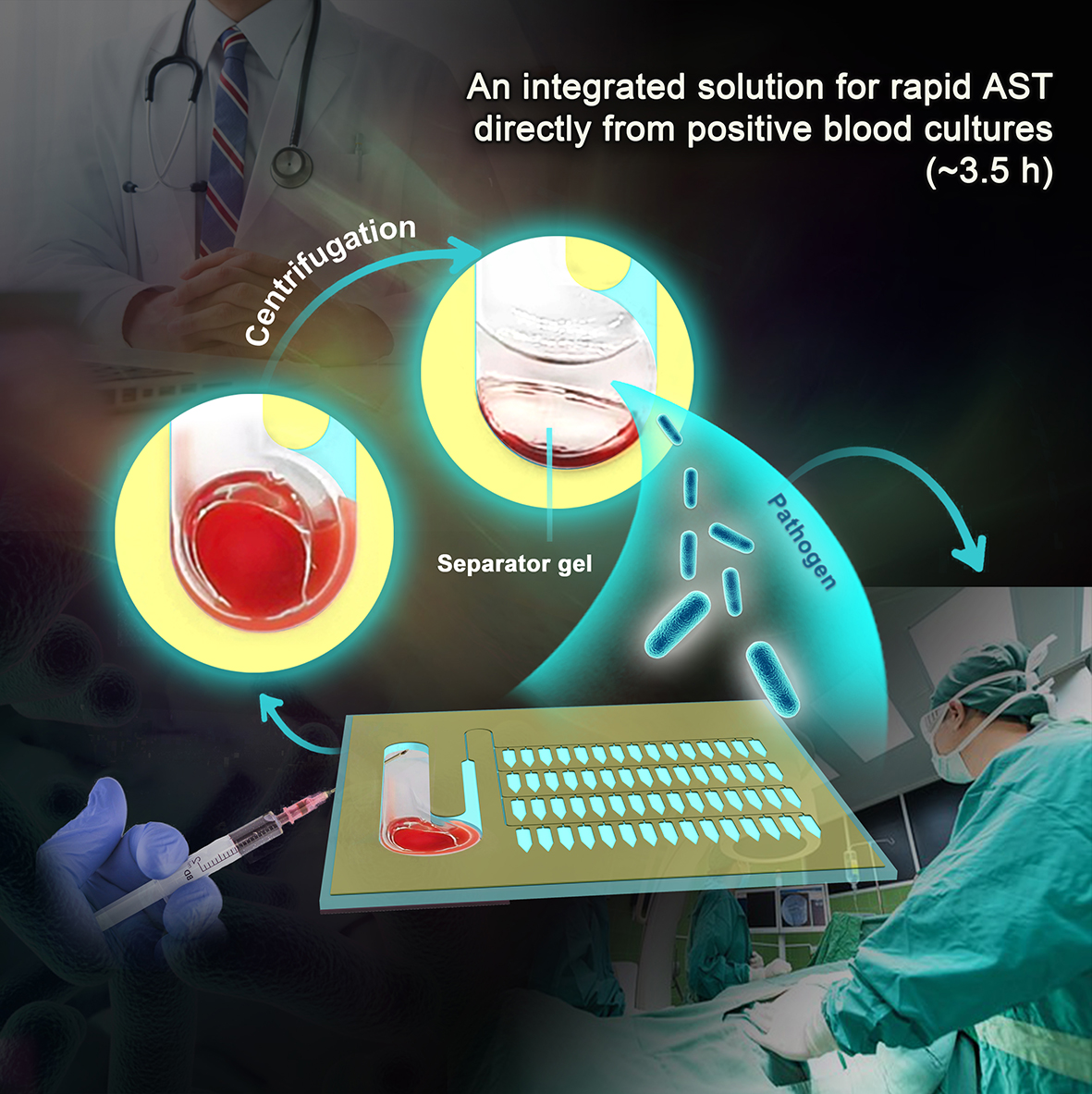
The on-chip pretreatment and rapid AST directly from positive blood cultures (Image by LIU Yang)
"Traditional AST methods currently require at least two days to yield results following a positive blood culture. The delay in diagnosis compels the administration of empirical antibiotics, risking the aggravation of the patient's condition and fostering the emergence of antibiotic resistance," said Prof. MA Bo from the Single-Cell Center at QIBEBT, co-author of the study. "Therefore, there is an urgent need for new technologies that can provide accurate and timely diagnostics and drug susceptibility testing."
In this study, the researchers designed a BSI-AST chip capable of extracting bacteria directly from PBCs within 10 minutes, and providing rapid AST results within three hours.
In a proof-of-concept study, the BSI-AST chip demonstrated its effectiveness by conducting direct AST on artificial PBCs containing E. coli, testing against 18 antibiotics. The whole process, from bacteria extraction to AST result output, required less than 3.5 hours.
Moreover, the integrated chip was applied to the diagnosis of clinical PBCs, showing a categorical agreement of 93.3% with standard methods in clinic. The reliable and rapid AST results of the integrated chip highlight its great potential in clinical diagnosis.
"In previous studies,microfluidic devices were mainly designed for purification and concentration of viable microorganisms derived from subculture or urine samples with simple composition," said ZHU Meijia, a doctoral student from Shandong University and first author of the study. "The practical utilization of these devices faced significant challenges due to the absence of on-chip complex sample preparation processes."
"The BSI-AST chip was developed for direct AST from PBCs without the need for subculture, marks a significant advancement," stated XU Teng, assistant research fellow and contributing author from Single-Cell Center at QIBEBT.
By introducing a separator gel to the microfluidic chip for the first time, the researchers achieved rapid extraction and enrichment of bacteria from PBCs. In combination with centrifugal microfluidic enrichment technology, AST results could now be obtained in less than 3.5 hours directly from PBCs. Furthermore, the demonstration of the chip's multiplexing analysis capability through antibiotic drying and array parallelization would support clinicians in promptly confirming or optimizing effective antibiotic therapy for BSI patients.
The introduction of the BSI-AST chip provides a rapid and convenient solution for sample pretreatment when combined with Clinical Antimicrobial Susceptibility Test Ramanometry (CAST-R), according to Prof. XU Jian, the head of the Single-Cell Center at QIBEBT.
"Rapid AST in blood culture is significant for patients with clinical sepsis and has the potential to save lives," emphasized Prof. CHENG Yongqiang, the corresponding authorfrom Shandong University. "Exploration and research in this field are expected to improve the diagnostic capabilities and provide valuable treatment guidance for infectious diseases in laboratory settings, effectively combating the serious threat of microbial resistance to humanity."